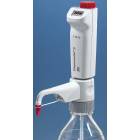
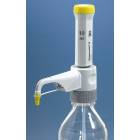
BrandTech Dispensette S Trace Analysis Bottletop Dispenser - Analog Adjustable with Standard Valve - Volume 1-10mL
Product Description
Dispensing of High-Purity Chemicals in Trace Analysis:
- The Dispensette® S Trace Analysis provides outstanding performance for precise volume dispensing of high-purity media for trace analysis. The components of the fluid path have been selected to only contain the highest purity materials, such as fluoroplastics and sapphire. Trace metal content of dispensed liquid generally in the low ppb range, or even in the low ppt range, depending on the application.
- Plastics in contact with media consist of high-purity materials such as PTFE, ETFE, PCTFE, FEP and PFA. The purest sapphire is used for replaceable valves. Depending on the application, Platinum-Iridium (suitable for dispensing HF) or Tantalum (well-suited for dispensing acids, bases and hydrogen peroxide) are available as spring materials.
- A field-tested cleaning process before use in trace analysis is described in the operating manual.
- The high-purity materials release virtually no metal ions after appropriate cleaning. This makes the Dispensette® S Trace Analysis bottletop dispenser a superior choice for trace analysis.
Features:
- Simple to mount discharge tube
- Designed without additional sealing rings for ease of cleaning and maintenance
- Replaceable discharge valve with safety valve
- Olive-shaped filling valve for firmer attachment of filling tubes
- Improved flow technology results in faster priming
- Scalloped track for secure volume setting on analog dispensers
- The 45 mm standard thread plus the included adapters fit most common lab bottles
- The valve block can be rotated 360° so that the bottle label always faces the user for safety
- Telescoping filling tube adjusts to different bottle sizes
- Easy to disassemble for replacement of the dispensing cartridge
The Dispensette S Trace Analysis Bottletop Dispenser with Standard Valve is supplied with a Valve Mounting/Calibration Tool, Telescoping Filling Tube, Discharge Tube, ETFE and PTFE Adapters, Operating manual, One-year warranty, and Certificate of performance.
Recommended Application Range:
- Platinum-Iridium (Pt-Ir) Valve Spring can be used for the following dispensing media: Acetic acid, Ammonia solution, Bromine, Hydrochloric acid, Hydrofluoric acid*, Nitric acid, Perchloric acid, Phosphoric acid, Sodium hydroxide 30 %, Sulfuric acid, Water
- Tantalum Valve Spring can be used for the following dispensing media: Acetic acid, Ammonia solution, Bromine, Hydrochloric acid, Hydrogen peroxide, Nitric acid, Perchloric acid, Phosphoric acid, Sulfuric acid, Water
- *Note: Hydrofluoric acid reacts slightly with sapphire resulting in mildly elevated aluminum values. To reduce these values we recommend discarding 3-5 dispensings of 2 ml each before performing the analysis. Compounds containing fluorine, such as NaF, attack tantalum.
Limitations of Use: This instrument is designed for dispensing liquids, observing the following physical limits:
- Use between +15°C and +40°C (59°F and 104°F) of instrument and reagent
- Density up to 3.8 g/cm³
- Vapor pressure up to max. 600 mbar; aspirate slowly above 300mbar in order to prevent the liquid from boiling
- Kinematic viscosity up to 500 mm²/s (dynamic viscosity [mPas] = kinematic viscosity [mm²/s] x density [b/cm³])
Operating Exclusions: Never use this instrument with:
- Liquids attacking Al2O3 sapphire or fluoroplastics like ETFE, FEP, PFA, PCTFA, and PTFE (e.g., dissolved sodium azide*)
- Liquids which are decomposed catalytically by platinum-iridium (e.g. H2O2) or tantalum, depending on the construction of the instrument
- Organic solvents
- Trifluoroacetic acid
- Explosive liquids (e.g., carbon disulfide)
- Suspensions as solid particles may clog or damage the instrument (e.g., of charcoal)
- The Dispensette® TA must NOT be autoclave
- *Dissolved sodium azide permitted up to a max. concentration of 0.1%
| Item# |
Volume (mL) |
Valve Spring |
Subdivision mL |
*A ≤ ± % |
*A ≤ ± µL |
*CV ≤ % |
*CV ≤ µL |
Supplied Adapter for Bottle Thread |
Supplied FillingTubeLength |
| 4640040 | 1-10 | Platinum-iridium | 0.2 | 0.5 | 50 | 0.1 | 10 |
GL28 (ETFE) GL32 (ETFE) S40 (PTFE) |
125-240mm |
| 4640240 | 1-10 | Tantalum | 0.2 | 0.5 | 50 | 0.1 | 10 |
GL28 (ETFE) GL32 (ETFE) S40 (PTFE) |
125-240mm |
*The value of Accuracy (A) and Coefficient of Variation (CV) are final test values referring to the delivered nominal volume, instrument and distilled water at equilibrium with ambient temperature (20°C/68°F) and with smooth operation.
Specifications
| Manufacturer | Brandtech Scientific |
|---|---|
| Latex Free | Yes |
| Country of Manufacture | Germany |
| Item Ships From | Connecticut |